Trudhesa Difference
Trudhesa® is the only migraine treatment that uses advanced Precision Olfactory Delivery (POD®) technology to consistently deliver dihydroergotamine mesylate (DHE) to the vascular-rich upper nasal space.2-4
DHE for the treatment of migraine has a 70+ year history of proven effectiveness1,5,6
DHE is recommended as a “Level A” acute treatment by the AHS Guidelines7,8
The efficacy of intravenous (IV) DHE is well-regarded by physicians because of its1,9:
- Rapid onset and sustained effects lasting up to 48 hours
- Efficacy irrespective of the time of treatment—from within 2 hours to beyond 4 hours, post migraine attack onset
- Efficacy against a full range of migraine symptoms, including pain, allodynia, photophobia, and phonophobia
In vitro studies show*:
- DHE can potentially reverse established central sensitization and allodynia1,9
- DHE has more prominent venoconstriction than vasoconstriction vs ergotamines10,11
*In vitro study data correlation to clinical outcomes are undetermined.
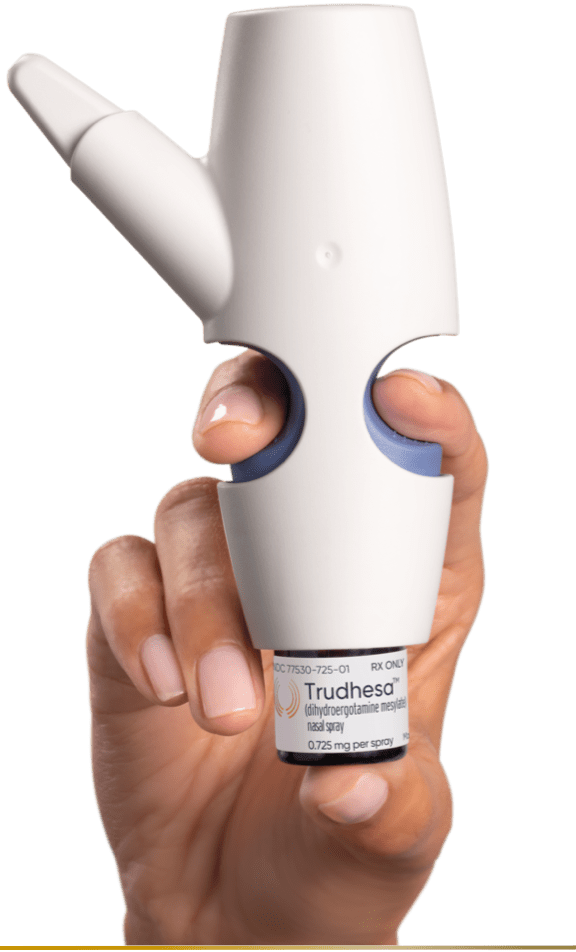
There’s nasal, and then there’s upper nasal12
The upper nasal space is highly permeable and vascular-rich, providing increased bioavailability by3,4,13:
- Consistent and predictable medication delivery (vs the significant drug loss common with lower nasal space delivery)
- Rapid absorption into the bloodstream
POD® technology
Traditional DHE nasal spray
POD technology—far from
just a nasal spray2
POD is manually actuated and propellant powered so coordinated inhaling is not needed2-4,12
- Gently and consistently delivers DHE into the upper nasal space
- Designed to prevent spilling down the front of the lip or down the throat to enable consistent drug delivery and possibly reduce adverse taste
- Avoids GI tract degradation and first-pass metabolism of oral treatments
- Circumvents the GI impact of autonomic dysfunction associated with migraine
Watch Dr. Wade Cooper discuss the difference between treating migraine with POD® vs traditional nasal sprays
Paid consultant of Impel Pharmaceuticals Inc.
TITLE: How can we treat migraine effectively through the nose?
Wade Cooper, DO: As a neurologist, we talk about the brain all the time, but for today, I want to focus on how cool the nose is and why it’s an important delivery for migraine medication.
When we talk about nasal anatomy, it’s important to know that all areas of the nose aren’t the same. The top of the nose, the back of the nose is really rich in both venous and arterial access. This, of course, is important because when you put medication in the anterior vestibule of the nose, it’s not ideal for medication absorption.
When we look at the lower nasal space, that anterior vestibule of the nose, it’s just not designed for medication absorption. It’s got squamous epithelium that acts almost like a raincoat and won’t let medication penetrate.
Now, if we focus on the upper nasal space, you’ll notice that the actual cells are different. These are columnar epithelial cells, and they’ve got cilia on them. But what’s interesting is that the cilia in the top of the nose are non-motile, which means that they don’t have a beating-like wave to move medication away.
And in my opinion, this is the most ideal area to deliver medications because these columnar epithelial cells are covered with mucus and able to absorb medication best for any spot in the nose.
If you can get medication to the top of the nose, where these blood vessels are ready to absorb medication, you’re going to get your optimal effect.
When we talk about the upper nasal space and why it’s so ideal for our migraine patients, we know that the upper nasal space is designed for phenomenal medication absorption. But we also know that medications that go to the upper nasal space avoid first-pass phenomenon in the gut and avoid you going through the liver to metabolize the medication down, and therefore giving you lower side effect options. And the third part about this is that medications, when they’re delivered in the upper nasal space, are also going to allow you to get past gut slowdown, which occurs when people have their acute migraine attacks.
So we know that traditional nasal atomizers, has a puff of medicine that goes into the front of the nose in what we call the nasal vestibule. And the reason why that medicine can’t go to the top of the nose is because of the portion of the nose called the internal nasal valve. The internal nasal valve isn’t an actual structure, it’s more of a stricture in the nasal passages, but it stops typical aerosolized medications from getting to the upper nasal space.
This becomes a huge barrier for delivering medicines to the top of the nose. And the POD technology with Trudhesa gets medicine past this internal nasal valve and up to the target where it’s going to be best absorbed.
Limitations of some oral treatments
Slow onset of action
- GI disorders are often associated with migraine and can result in delayed absorption and bioavailability14-16
- Oral treatments may not be fully absorbed due to migraine-associated GI symptoms14-16
Trudhesa circumvents GI issues by rapidly and consistently delivering DHE to the vascular-rich upper nasal space and into systemic circulation.2,4,17
Limitations of some oral treatments

Limited dosing windows
- Some oral acute treatments need to be taken within 60 minutes of migraine attack onset to be most effective18
- Many patients delay taking treatment for several reasons19†
Trudhesa can provide relief even when taken late into a migraine attack.1,3,9‡
†In a US study of 690 patients where 338 patients often avoid or delay taking migraine medication, common reasons for delaying treatment were waiting to see if it was a migraine attack, only want to take medication for a severe attack, and concerns about side effects.19
‡Based on a phase 3, open-label, safety study evaluating exploratory efficacy outcomes and post hoc analyses.3

Lack of consistency and sustained response
Migraine-associated GI issues may impact complete and consistent absorption of oral treatments20-23
- Studies of oral triptans reported the incidence of recurrence varied from 6% to 50%
- In studies of gepants, sustained pain freedom 24 hours postdose was achieved by about 13% to 16% of patients
Trudhesa provides consistent delivery of proven long-lasting DHE, for sustained relief time after time.2-4
of patients do not respond to treatments like triptans and gepants that act on singular receptor types in the trigeminovascular system24
References: 1. Silberstein SD, Shrewsbury SB, Hoekman J. Dihydroergotamine (DHE) – then and now: a narrative review. Headache. 2020;60(1):40-57. 2. Trudhesa. Prescribing information. Impel Pharmaceuticals; 2021. 3. Smith TR, Winner P, Aurora SK, Jeleva M, Hocevar-Trnka J, Shrewsbury SB. STOP 301: a phase 3, open-label study of safety, tolerability, and exploratory efficacy of INP104, Precision Olfactory Delivery (POD®) of dihydroergotamine mesylate, over 24/52 weeks in acute treatment of migraine attacks in adult patients. Headache. 2021;61(8):1214-1226. 4. Shrewsbury SB, Jeleva M, Satterly KH, Lickliter J, Hoekman J. STOP 101: a phase 1, randomized, open-label, comparative bioavailability study of INP104, dihydroergotamine mesylate (DHE) administered intranasally by a I123 Precision Olfactory Delivery (POD®) Device, in healthy adult subjects. Headache. 2019;59(3):394-409. 5. US Food and Drug Administration. Drugs@FDA: FDA-Approved Drugs. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm. Accessed August 24, 2021. 6. Saper JR, Silberstein S, Dodick D, Rapoport A. DHE in the pharmacotherapy of migraine: potential for a larger role. Headache. 2006;46(suppl 4):S212-S220. 7. Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: The American Headache Society evidence assessment of migraine pharmacotherapies. Headache. 2015;55(1):3-20. 8. American Headache Society. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59(1):1-18. Published correction appears in Headache. 2019;59(4):650-651. 9. Aurora SK, Ray S, Satterly K, Shrewsbury SB, Hoekman J. Does dihydroergotamine treat the “whole migraine”? Poster presented at: American Headache Society Virtual Annual Scientific Meeting, June 2020. 10. Lipton RB. Ergotamine tartrate and dihydroergotamine mesylate: safety profiles. Headache. 1997;37(suppl 1):S33-S41. 11. Saper JR, Silberstein SD. Pharmacology of dihydroergotamine and evidence for efficacy and safety in migraine. Headache. 2006;46(suppl 4):S171-S181. 12. Martin V, Hoekman J, Aurora SK, Shrewsbury SB. Nasal delivery of acute medications for migraine: the upper versus lower nasal space. J Clin Med. 2021;10(11):2468. 13. Data on File. Impel Pharmaceuticals. 2020. 14. Aurora SK, Shrewsbury SB, Ray S, Hindiyeh N, Nguyen L. A link between gastrointestinal disorders and migraine: insights into the gut-brain connection. Headache. 2021;61(4):576-589. 15. Lipton RB, Munjal S, Buse DC, et al. Unmet acute treatment needs from the 2017 Migraine in America Symptoms and Treatment Study. Headache. 2019;59(8):1310-1323. 16. Axert. Prescribing information. Janssen Pharmaceuticals; 2017. 17. Aurora SK, Papapetropoulos, Kori S. Gastric stasis in migraineurs: etiology, characteristics, and clinical and therapeutic implication. Cephalalgia. 2013;33(6):408-415. 18. Lantéri-Minet M, Mick G, Allaf B. Early dosing and efficacy of triptans in acute migraine treatment: the TEMPO study. Cephalalgia. 2012;32(3):226-235. 19. Foley KA, Cady R, Martin V, et al. Treating early versus treating mild: timing of migraine prescription medications among patients with diagnosed migraine. Headache. 2005;45(5):538-545. 20. Láinez MJ, García-Casado A, Gascón F. Optimal management of severe nausea and vomiting in migraine: improving patient outcomes. Patient Relat Outcome Meas. 2013;4:61-73. 21. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant for the treatment of migraine. N Engl J Med. 2019;381(23):2230-2241. 22. Croop R, Goadsby PJ, Stock DA, et al. Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial. Lancet. 2019;394(10200):737-745. 23. Diener HC, Tassorelli C, Dodick DW, et al; International Headache Society Clinical Trials Standing Committee. Guidelines of the International Headache Society for controlled trials of acute treatment of migraine attacks in adults: Fourth edition. Cephalalgia. 2019;39(6):687-710. 24. Barbanti P, Egeo G, Mitsikostas DD. Trigeminal-targeted treatments in migraine: is 60% the magic number? Headache. 2019;59:1659-1661.

